purpose
The demonstration uses Empower ® 3 ICH impurity treatment to simplify the analysis of impurities in ziprasidone hydrochloride by defining the allowable threshold limits and quickly identifying results beyond the limits.
background
Impurity analysis, including identification and quantitative analysis, of pharmaceutical and pharmaceutical products is a requirement that every manufacturer must follow. Therefore, rapid and accurate assessment of impurities generated by active pharmaceutical ingredients (APIs) in pharmaceutical product formulations and development processes is required. The presence of impurities can affect the safety and efficacy of the final drug product and must be effectively monitored. The International Conference on Harmonization (ICH) published guidelines for impurities in Drugs 1 and 2 , providing an allowable threshold limit for impurities to monitor the safety of drugs and pharmaceutical products.
The Empower 3 ICH Impurity Processing feature simplifies quantitative analysis by quickly identifying user-defined impurities that exceed the ICH allowable limits. Empower software automatically compares the calculated content of impurities to the limits and marks the results of the failure. This allows rapid assessment of the safety of the drug product during formulation or release testing.
Empower 3 ICH Impurity Processing allows the user to define an allowable threshold limit for impurities and quickly identify if the result exceeds the limit.
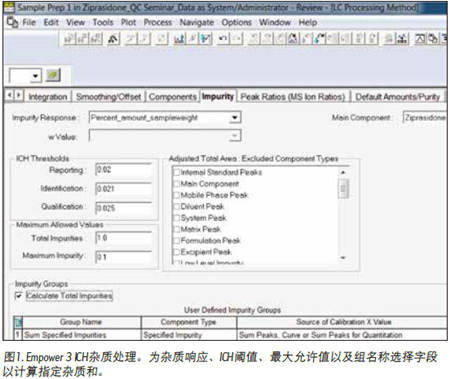
This technical brief demonstrates the use of Empower 3 ICH impurity treatment to simplify the quantitative analysis of impurities in ziprasidone hydrochloride in capsule preparations.
solution
Waters Application Note 720004639ZH previously developed a UPLC method for ziprasidone hydrochloride and its related substances specified in USP 3 . Quantitative analysis of the relevant substances in the ziprasidone capsule preparation was simplified by using Empower 3 ICH impurity treatment to define ICH threshold limits and to identify results that did not exceed the limits. First, we specified the formula for calculating the amount of impurities or impurity response in the Empower processing method (Figure 1). Next, we define the ICH threshold for reporting, identifying, and characterizing impurities. Then, the maximum allowable value is specified for total impurities and maximum impurities. In this example, we used a more stringent standard than the ICH guidelines to demonstrate the workflow for identifying and labeling peaks that exceed the impurity threshold limits. We used a reporting threshold of 0.02% API, which is less than 0.1% of the reported impurity value in new drug products as specified in the ICH guidelines (based on maximum daily dose ≤ 1g) 2 . In addition, we can select tighter limits than the general ICH threshold for each of the specified impurities in the drug product. In addition, we can group specific types of impurities to calculate their total amount.
Finally, the chromatographic data is processed to determine the total amount of impurities. The data (Figure 2A) shows that Compounds A and B were detected in the capsule samples using the Empower patented Peak Detection ApexTrackTM integration algorithm. Empower software quickly identified Compound A's over-the-counter (OOS) results (marked in red) because this value exceeded the threshold limit defined in the processing method (Figure 2B). In addition, the analysis of impurities can be easily reported (Figure 3). The report template can be customized to meet the reporting needs of each user, and the electronic signature function can be conveniently used, as shown in Figure 3.
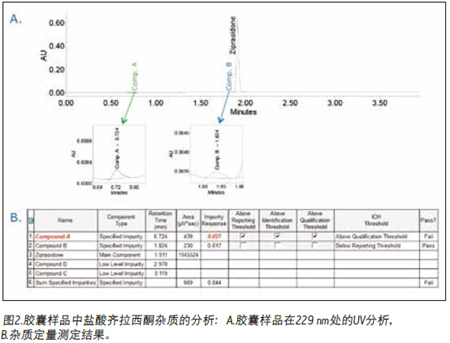
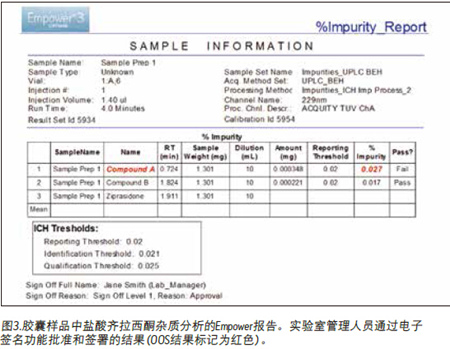
to sum up
Empower 3 ICH Impurity Processing allows the user to define an allowable threshold limit for impurities and quickly identify if the result is within limits. Empower clearly displays user-defined threshold limits during data review or reporting. This reduces time-consuming, reduces potential errors associated with manual verification, and increases confidence that any results that exceed the threshold limit can be quickly identified. Overall, Empower 3 ICH Impurity Treatment is an easy-to-use yet powerful tool for any laboratory to monitor impurity levels during pharmaceutical product preparation or finished product release testing, while simplifying the quantitative analysis program.
references
1. ICH Q3A (R2), Impurities in New Drug Substances, International Conference on Harmonization,
October 2006.
2. ICH Q3B (R2), Impurities in New Drug Products, International Conference on Harmonization, June 2006.
3. Maziarz M, Jones MD, Potts WB, Forini F. Transfer of Two UPS Compendial Methods for Impurities of Ziprasidone HCl to a Single UPLC Method. Waters Application Note 720004639EN, April 2013.
We're Professional Supplier Extract Powder manufacturers and suppliers in China specialized in providing high-quality products at low price. We warmly welcome you to buy or wholesale bulk Supplier Extract Powder for sale here from our factory. For a free sample, contact us now.
Supplier Extract Powder,Supplier Extract ,Supplier Powder Manufacturer in China
Shaanxi Kang New Pharmaceutical co., Ltd. , https://www.apipepdite.com