Release date: 2016-06-30
Things happened in mid-June, and the FDA approved a new weight-loss device. However, some doctors have objected to this, and one of the doctors even plans to sue FDA agencies together with 4,000 doctors. The reason for this is that doctors believe that this device, which pumps the food just taken by the patient directly from the stomach to achieve weight loss, has a great safety hazard and may also lead to eating disorders.
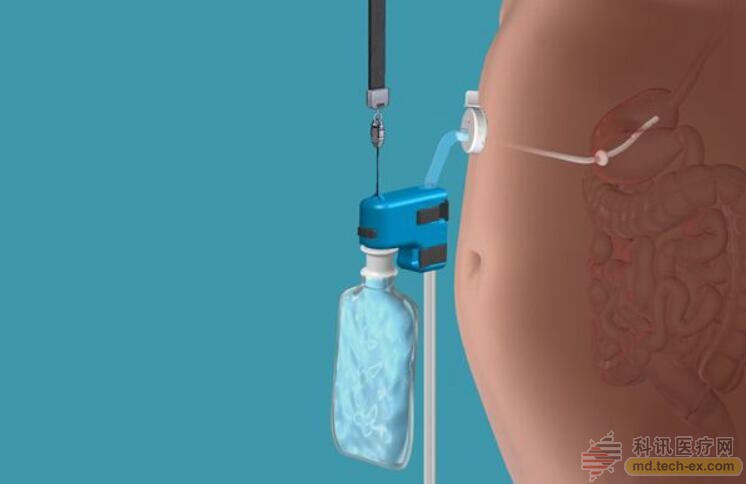
The schematic of the use of this new weight loss device
Joseph Gutman, an endocrinologist and diabetes expert with more than 30 years of experience in obesity treatment in Pembroke Pines, Fla., said, "In my long career, this was my first FDA-approved device. Spectral, I can't believe that this device will be approved.†Gutman said that he has now convened 750 doctors who plan to sue the FDA, and his ultimate goal is to call 4,000 doctors to join their team. They hope that this device will be banned from being placed on the market. They believe that this approval is an FDA's ignorance of clinical medicine. It is just a bad little trick, and it is like a boring joke that will have serious consequences.
What exactly is this new weight-loss device?
The device, called AspireAssist, was approved by the FDA after 111 years of clinical trials by 111 researchers. The FDA noted that the device targets the obese patients with a body mass index (BMI) ranging from 35 to 55 and who use non-surgical therapy to lose weight (BMI is a measure of whether a person is obese in terms of overall height and weight. Index; a person 5 feet 9 inches tall, if the BMI is in the 35 to 55 range, the weight is about 236 to 365 pounds after conversion). According to the FDA, AspireAssist is not available to patients with eating disorders.
During the use of the AspireAssist, a catheter is inserted through the abdomen into the patient's stomach. It is accessed through a "port valve" that is located directly above the patient's navel. The patient can open and close the valve to expel food from the stomach. 20 to 30 minutes after the meal, the patient can connect the pump to the valve, and the appetite in the stomach can be exported and discharged directly into the toilet. This process takes about 10 minutes.
It is reported that AspireAssist is developed by Aspire Bariatrics, based in Prussia, the capital of Pennsylvania. According to Kathy Crothall, CEO of the company, this method of direct food intake from the stomach can reduce the intake of 30% of the total calories in each meal. .
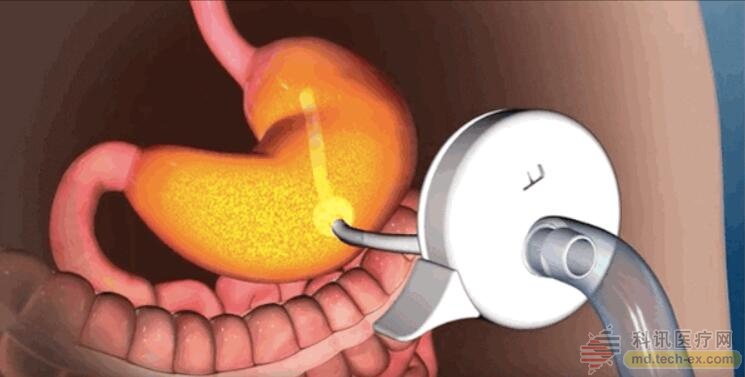
AspireAssist drains food directly from the stomach
Experts' concerns
1, may lead to patients with eating disorders
However, in the opinion of some experts, AspireAssist is accompanied by some side effects, such as the port valve can cause infection and the catheter may leak. However, what most experts worry about is that it may lead to eating disorders in patients. This is because the working mode of the device is similar to the symptoms of binge eating disorder in a certain sense, that is, uncontrolled overeating, and then vomiting food, and many people with obesity are also binge eating patients. “An eclipse vomits food from the throat and AspireAssist is drained through the catheter. This is a mechanized bulimia patient, and it also indulges an ecstasy patient from another outlet to vent their appetite,†Gutman explains.
Aspire Bariatrics said that it is impossible for gluttons to vent their gluttony syndrome through AspireAssist. Because the food must be chewed completely before it can be expelled through the catheter. At the University of Washington in St. Louis, Shelby conducted a clinical trial of AspireAssist on 17 devices. Sullivan said that if the patient does not chew the food completely, the oversized restaurant will get stuck in the catheter and cannot be discharged. At the age of 52, Lotta Frisk lost 148 pounds after using AspireAssist for four years. Every time she eats, she chews 55-75 times of every bite. Lotta Frisk said, “I can't just swallow the food without chewing, and then immediately discharge it. I need to chew it many times before the food can be discharged through the AspireAssist catheter.â€
There are also clinical studies that show that patients who use this weight loss device need to chew food several times while eating, so the longer they eat, the stronger their satiety, and the less food intake. This also achieved the effect of weight loss to a certain extent.
Eric Wilcoxon, 44, has been using AspireAssist since 2013 and she agrees with this reduction in food intake. Because she needs to chew many times, when she starts eating less meals, she chooses to eat fruits, steamed vegetables, grilled chicken, etc., which are easy to chew and easy to absorb. "If you can't understand the importance of chewing, all you can do is to chew your food carefully. After you develop this kind of eating habit, you will understand." Wilcoxon said.
As of now, Wilcoxon, which weighed 389 pounds, has lost 128 pounds with the help of AspireAssist. Three years ago, when Wilcoxon gave up weight loss surgery and defeated the fear of complications from the use of weight loss devices, he decided to pick up the device at the Clinical Trial Center at the University of Washington. Previously, he had seen good friends lose weight by doing gastric banding surgery, which would shrink the stomach to the size of a golf ball to limit the patient's food intake. Compared to this bariatric surgery, it is clear that AspireAssist is more attractive to Wilcoxon. In addition, he plans to use the weight loss device for life, because this weight loss method still allows him to enjoy the favorite food.
In addition, chewing is not the only behavioral change that occurs during the use of AspireAssist. Studies have shown that people who use the device will choose healthier foods, and they actually see the form of the chyme that is discharged from their stomachs, so they will repeatedly consider the food they eat. Users said that healthy food turned into a restaurant that looked less disgusting, similar to when it was eaten, but was broken down into the stomach. But junk foods such as burgers and French fries seem to be very disgusting when they are discharged from the catheter.
2, worry that this weight loss device will bring long-term sequelae to patients
AspireAssist only had a one-year clinical trial period before being approved by the FDA. This has led some experts to worry about whether they will have some long-term aftereffects. "Although in the long run, there is no scientific basis to judge what side effects it will bring," Gutman said. "But the study is not perfect. It is also difficult to know how it will react once patients stop using the device. Will their weight rebound immediately?" Raul, president of the American Society for Metabolism and Surgical Weight Loss Rosenthal believes that if a patient suddenly discontinues after using AspireAssist for one to two years, 98% of patients will bounce back to their original weight. This is because most people can't control it and start unhealthy eating habits.
Some scholars worry that this type of weight-loss device will give patients the illusion that they can eat without any scruples, no matter how much they eat, they can use this mechanized way. However, the true scientific treatment of obesity means that eating habits should be changed. Patients should be advised to eat less and to eat healthy foods in conjunction with exercise. “It’s hard for me to accept this seemingly simple approach, and it also allows patients to eat more and not absorb calories to lose weight,†says Konstantinos Spaniolas, a professor at the University of East Carolina School of Medicine.
Previously, during the FDA's review of its clinical trials, participants used AspireAssist along with a number of nutrition and exercise counseling. On average, they lost 12% of their initial weight, while those who only received nutrition and exercise counseled lost an average of 4%. Although it seems better to use AspireAssist's weight loss effect on the surface than just diet control and exercise, when we consider the process of invading the human body into consideration, we feel that this result is not so satisfactory, Spaniolas said. . During the clinical trial, the AspireAssist insert process took 15 minutes but did not perform general anesthesia. Both Frisk and Wilcoxon said that they were able to adapt to the pain when they put the catheter into the stomach for the first two weeks. The FDA also warned them that this process carries the risk of stomach infections, bleeding, vomiting, nausea, sores and other symptoms.
In fact, surgical bariatric surgery can help obese patients achieve better weight loss. For example, sleeve-shaped gastrectomy removes 80% of a person's stomach, which can help patients lose about 30% of their weight; gastric bypass surgery allows food to "pass" most of the stomach and directly into the intestines, which can help The patient lost about 35% of his weight. But these two procedures are irreversible and have the potential to cause serious complications.
“For obese patients, a 12% weight loss is a good goal. But this device invades the body and introduces a tube into the stomach, so it is directly connected to the skin and stomach. In my case, it's not like a safe and effective way to treat disease, but it sounds more like an adventurous game,†Spaniolas said.
In fact, the treatment of obesity is not only done by weight loss, but also has many advantages in traditional surgical treatment. Just after weight loss surgery may lead to some complications, such as diabetes, high blood lipids, high blood pressure, or changes in hormone levels.
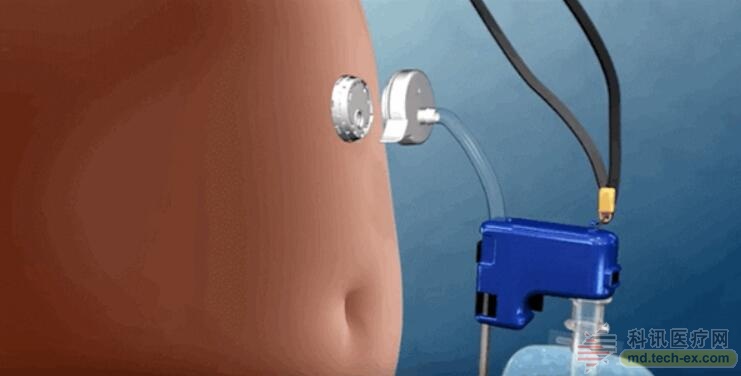
In 2012, Frisk, who was still living in Sweden, got AspireAssist, who weighed 320 pounds and had high blood pressure, type 2 diabetes and depression. For patients like Frisk, losing weight is enough to improve their health. She uses it once a day and now she has lost 172 pounds. Frisk said that she did not take any medications to treat high blood pressure and diabetes but remained healthy. At the same time, depression has also improved a lot. She no longer feels depressed and sad every day, and there is no hope in life. Frisk, who currently lives in Delray Beach, Florida, believes that “I feel that I am really reborn, everything has changed, and I am happy every day.â€
500 people around the world use AspireAssist
Aspire Bariatrics CEO Crothall said that about 500 people around the world have used their equipment and proved to be safe and effective. (AspireAssist is already in use in Europe and plans to expand the market to Australia, Canada, New Zealand, South America soon). The device is very similar to the catheters we've been using for decades. AspireAssist just exports food instead of entering it, but the concept of the two is the same. And they firmly believe that this catheter stays in the abdomen for a period of time and does nothing to the human body.
But such rhetoric is not enough to convince Gutman, who are actively preparing for legal aid to sue FDA doctors. They agreed that AspireAssist can cause various complications, such as infection, bleeding, and leakage. Gutman also wrote a complaint letter to the FDA. He said, "I don't think there is any logic about this kind of equipment. I don't think it has any medical or physiological significance. It's ridiculous than Trump's campaign for the president."
Today, more than 79 million people in the United States suffer from obesity, accounting for 35% of the total population. Therefore, it is crucial and urgent to develop some new equipment and put it into the market, and to propose more treatment methods for patients to choose. But every new device takes time to prove whether it really works. Rosenthal, a member of the American Society for Metabolic and Surgical Weight Loss, said objectively, "In fact, approval by the FDA only means that a device or drug is considered safe, but should never be considered the best treatment. In other words, even if it is approved by the FDA, it does not mean that the therapy can be applied to every patient."
Source: Arterial Network
China Extract Powder For Use As Dietary Supplement Extract Powder, Extract Powder Manufacturer
Shaanxi Kang New Pharmaceutical co., Ltd. , https://www.bodybuildingoil.com