Recently, Huahai Pharmaceutical received a notice from the US FDA, and the company's application for a new drug for rivaroxaban tablets (ANDA, a US generic drug application) has been temporarily approved (temporary approval) The FDA has completed all review requirements for generics, but is granted a form of approval due to the expiration of patent or monopoly rights. Up to now, Huahai Pharmaceutical has invested in research and development expenses of about 147.05 million yuan on the Livosa tablet project.
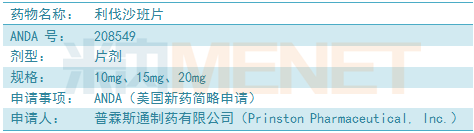
Rivaroxaban tablets are mainly used to treat adult patients undergoing elective hip or knee replacement surgery to prevent venous thrombosis (VTE).
Sales of rivaroxaban tablets in public medical institutions in China (unit: 10,000 yuan)
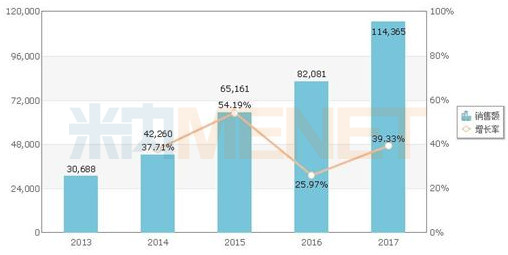
Because the drug is still in the patent protection period, only the original research products are on the market. According to the data from the intranet, in 2017, the sales of rivaroxaban tablets in public hospitals (urban public hospitals , county-level public hospitals, urban community centers and township hospitals) exceeded 1 billion yuan, a year-on-year increase of nearly 40%. The manufacturer is only Bayer. The main sales channel is in urban public hospitals, accounting for more than 90%.
Up to now, Huahai Pharmaceutical has invested in research and development expenses of about 147.05 million yuan on the Livosa tablet project.
Huahai Pharmaceutical said that this rivarshaban tablet received the FDA temporary approval number, which indicates that the product has passed all the review requirements of generic drugs. However, the product needs to be eligible for sale in the US market after the patent expires and is finally approved by the FDA. The FDA's provisional approval number for this rivaroxaban film marks the further advancement of the company's business in the field of generic products, which has a positive impact on the company's expansion into the US market.
Rice net white feather finishing
Source: Listed company announcement, Minenet database
41022 Green Tea,Green Tea 41022,Azawad Tea Alfata Ould Mohamed,Zahra Tea Nouveau Soleil Sarl
DIYAS DEVELOPMENT LIMITED , https://www.diyastea.com