Significantly reduce the number of seizure days, migraine new drug phase 3 clinical results are positive
June 12, 2018 Source: WuXi PharmaTech
Window._bd_share_config={ "common":{ "bdSnsKey":{ },"bdText":"","bdMini":"2","bdMiniList":false,"bdPic":"","bdStyle":" 0","bdSize":"16"},"share":{ }};with(document)0[(getElementsByTagName('head')[0]||body).appendChild(createElement('script')) .src='http://bdimg.share.baidu.com/static/api/js/share.js?v=89860593.js?cdnversion='+~(-new Date()/36e5)];Allergan today announced that its ongoing drug, Atogepant, has achieved positive results in clinical phase 2b/3 trials, reaching the primary study endpoint in all dose regimens and significantly reducing migraine in a statistically significant manner. The number of intermittent monthly episodes of the patient. Atogepant is a novel oral calcitonin gene-related peptide (CGRP) receptor antagonist.

Migraine is a chronic disease with neurological symptoms such as headache, sensitivity to light, sound, and nausea. These symptoms are usually incurable and can last for more than a day, seriously hampering the patient's normal life function. It even caused the work to be completely stagnant. Migraine is very common, affecting about one-seventh of the world's population (affecting 12% of the US population, including children) and is associated with significant disability, leading to social and economic burdens. Patients with intermittent migraine usually have 4-14 days of headache per month. Current care standards for acute treatment of migraine are not optimal for many patients, and there are limitations such as limited efficacy, poor tolerance, or contraindications. Patients may experience uncontrolled migraine repeatedly, leading to increased drug overuse and increased risk of disease progression. Compared with current standards of care, new migraine treatments are needed to increase effectiveness, reduce risk, and benefit more patients.
CGRP and its receptor are expressed in the nervous system region associated with the pathophysiology of migraine. CGRP receptor antagonism is a new mechanism of action in the acute treatment of migraine, which is significantly different from the existing triptans (serotonin 1B / 1D agonist) and opioids. Atogepant is the second CGRP receptor antagonist developed by Ai Jian following Ubrogepant, which can significantly reduce the number of days of acute migraine attacks. Ai Jian believes that CGRP antagonist drugs can complement the company's Botox, which can be used to treat chronic migraine.
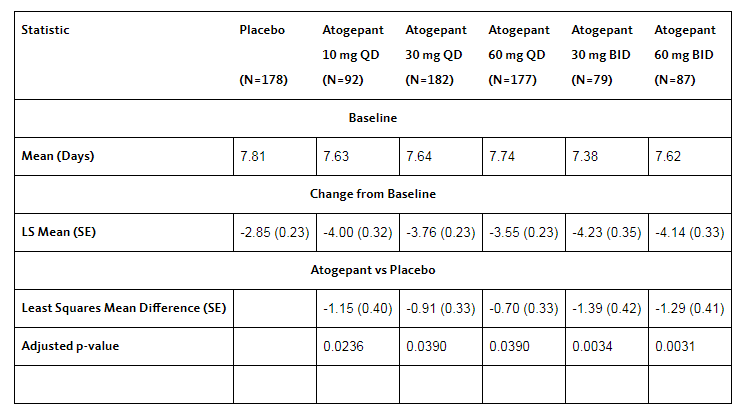
â–²CGP-MD-01 clinical trial results (Source: Ailjian official website)
The CGP-MD-01 clinical trial was a multicenter, randomized, double-blind, placebo-controlled, parallel-group study of phase 2b/3 in the United States for a period of 12 weeks. The trial included a random 4-week screening/baseline period, a 12-week double-blind treatment period, and a 4-week safety follow-up period. 834 subjects aged between 18 and 75 years (mean 40.1 years), history of migraine (with or without aura) for at least 1 year, and baselines were screened 3 months and 28 days prior to the first visit There were an average of 4 to 14 migraine/most likely migraine (MPM) episodes per month during the period. A total of 82% of patients completed the study, and the discontinuation rate was similar in all treatment groups. The results of the trial showed that Atogepant reached the primary endpoint in all dose regimens. Efficacy analysis was based on a population of 795 modified intention-to-treat (mITT) patients. The primary efficacy endpoint was the change in mean monthly MPM episodes from baseline over the 12-week treatment period. In addition, Atogepant was well tolerated and liver safety was similar to the placebo group.
Dr. David Nicholson, Executive Vice President and Chief R&D Officer of Alfred, said: “Ai Jian is very satisfied with the results of this 2b/3 trial. This reflects Ai Jian’s commitment to developing migraine preventive therapy. More data on the end of the trial is expected to be released at future scientific conferences. Over the past few decades, we have been working on new treatments for migraine headaches. We are committed to solving migraine patients through innovative drugs that have not been met. The need for treatment, they are looking forward to new treatment options that will bring real changes to their daily lives."

â–² Mr. Bill Meury, Chief Commercial Officer of Aerjian (Source: Ai Jian official website)
Mr. Bill Meury, Chief Commercial Officer of Aerjian, said in a statement: "The positive results of this study show that the oral results of Atogepant are convincing compared to other therapies on the market and the means of preventing migraine. We are excited about the prospects of this product and will move quickly to the next stage of development. Aijian has one of the most innovative and in-depth migraine product lines in the industry, of which Botox has been approved for the prevention of chronic migraine, oral Atogepant and Ubrogepant are in the development phase for the treatment and prevention of acute exacerbations of migraine."
We hope that this new migraine drug can be applied for market as soon as possible, which can help the majority of migraine patients to prevent migraine.
Reference materials:
[1] Allergan's Atogepant Hits the Mark in Phase IIb/III Episodic Migraine Trial
[2] Allergan's Oral CGRP Receptor Antagonist Atogepant Demonstrates Robust Efficacy and Safety in Episodic Migraine Prevention in a Phase 2b/3 Clinical Trial
[3] Treatment of migraine, the key clinical phase of new drug success
[4] Significantly reduce symptoms, migraine new drug phase 3 clinical results are positive
Portable Horizontal Band Sawmill
Shandong Longze Mechanical Equipment Co.,Ltd , https://www.pelletmachinefactory.com