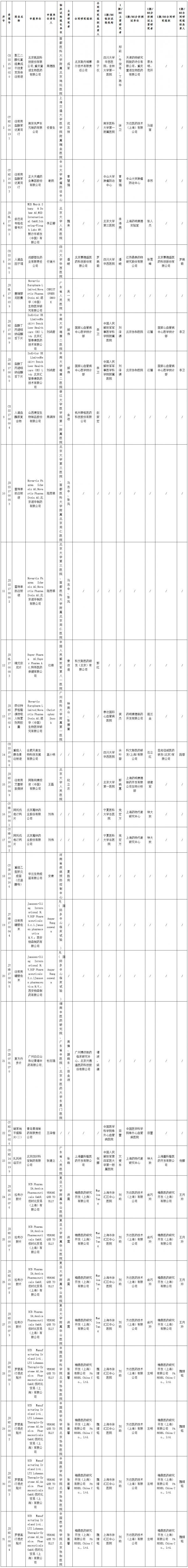
Recently, the Food and Drug Administration and Inspection Center of the State Food and Drug Administration issued the Announcement on the On-site Verification Plan for Drug Clinical Trial Data (No. 13).
Announcement: According to the "National Food and Drug Administration Drug Clinical Trial Data Verification Work Procedure (Provisional)" (Food and Drug Administration Chemicals [2016] No. 34), plans to PEGylation recombinant integrated interferon variant injection The liquid drug (acceptance number: CXSS1500020) and other 31 clinical trial data of the drug self-checking and verification varieties (see attachment) for on-site verification, is now publicized.
The publicity period is 10 working days, from August 18, 2017 to August 31, 2017. After the end of the publicity period, on-site verification will be carried out.
Our mission: To protect the health of ear, throat and nose with medical frontier knowledge and technological innovation.
Our vision: Leader in several niche markets of Otolaryngology.
Our values: Change, Enterprising, Share
Silicone Enema Bag, Enema Bulb, Enema Bucket, Automatic Enema Device
Ningbo Jiamai Internet Technology CO., Ltd. , https://www.jmcuhyd.com